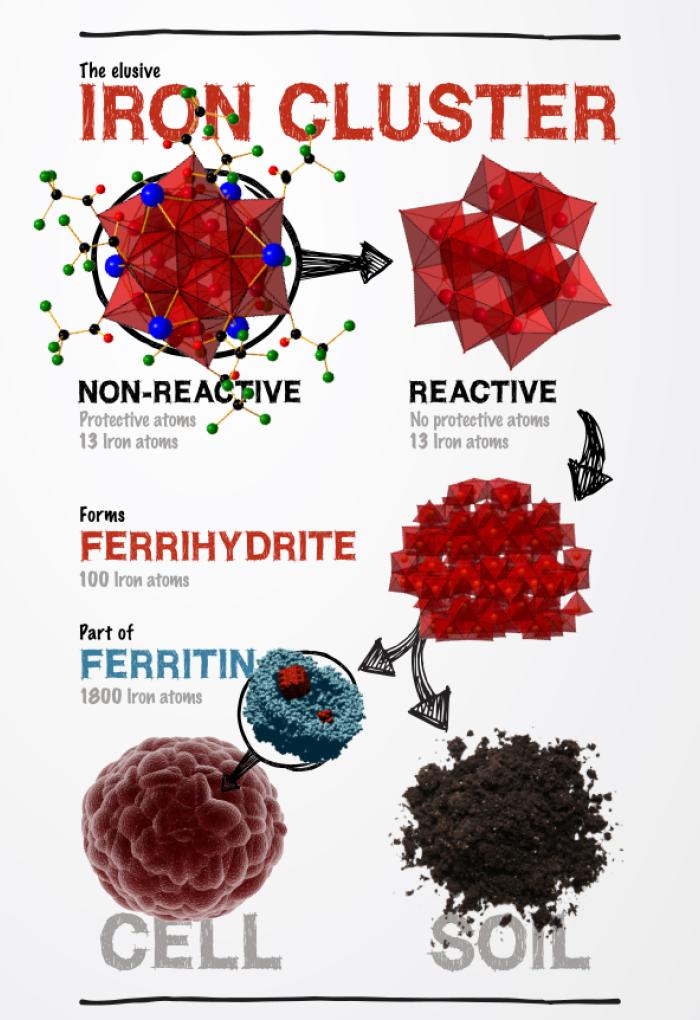
Chemistry researchers have successfully isolated a much-sought after iron compound—a 13-Fe atom cluster that is a prototype of ferrihydrite—the most abundant form of iron in natural systems, including our water, soil and the human body. Although scientists have long known the iron oxide cluster might exist, they could not study it because they couldn’t isolate it.
Due to its high negative charge and reactivity in water, this ‘crypto-cluster’ has proven extremely elusive in an isolated form. That is, until now.
Oregon State Associate Professor of Chemistry May Nyman and her team were able to capture the iron cluster—known as the Keggin ion, Fe13—in a discrete, stable form.
These findings were published in Science magazine today and supported by funding from the US Department of Energy’s Office of Basic Energy Sciences in the Division of Materials Sciences and Engineering. Lead author Omid Sadeghi, a second-year doctoral student from Iran, conducted much of the experimental in collaboration with Lev N. Zakharov, director of the University of Oregon’s X-ray infraction lab, and Nyman.
Here’s how it works: Nyman’s team used “protecting chemistry,” or strategically placed counterions to balance the high negative charge and temporarily mask the characteristic highly reactive chemistry. These atoms form a protective shell and stabilize the iron cluster. Without this protective shell, ferrihydrite forms immediately, preventing scientists from fully understanding how it actually forms. With additional controlled chemistry, Nyman’s team successfully removed the protective atoms, partially or fully, to track the conversion of the iron cluster to ferrihydrite.
The significance and impact of this breakthrough is profound: By isolating the iron oxide building blocks, scientists can gain an in-depth understanding of how ferrihydrite and magnetite form in nature and, notably, ferrihydrite’s role in iron regulation within the body.
Ferrihydrite serves vital roles in the interrelated events of contaminant transport, pH control of surface and ground water and microbial activity. As a result, this discovery may have major implications in our health, food supply and agriculture as well as bioremediation.
- First, knowing how iron is processed in our bodies can illuminate in the role iron plays in microbial and cellular processes, leading to more effective treatment of conditions like anemia.
- Second, a better understanding of iron in our soil can aid agricultural scientists to understand its influence on the environment, especially in the related physical, chemical and biological processes.
- Next, the ability to study these isolated forms of iron oxide can also bolster bioremediation efforts by pinpointing the role the element plays in removing or neutralizing pollutants and hazardous substances from natural and potable waters, such as pharmaceutical or radioactive biohazards.
- Perhaps most notably, iron is a less toxic and cheaper way to clean water, compared to using aluminum to purify our waters supplies.